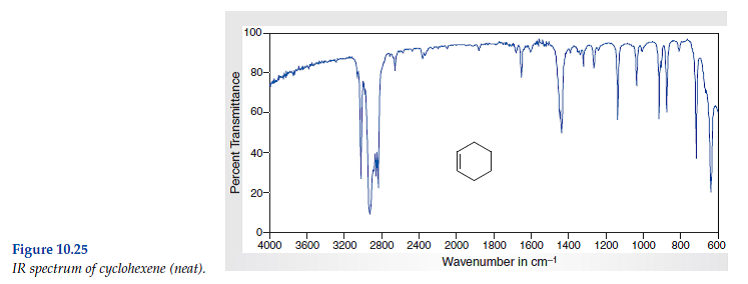
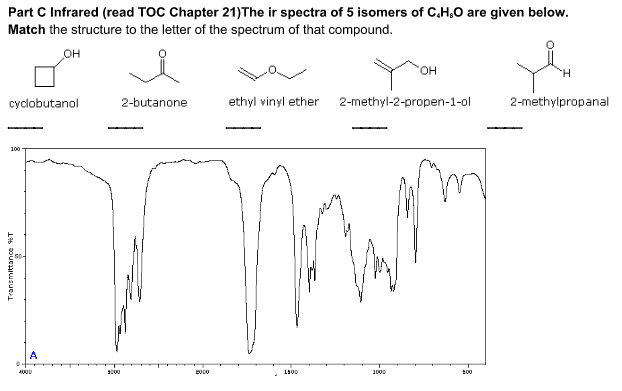
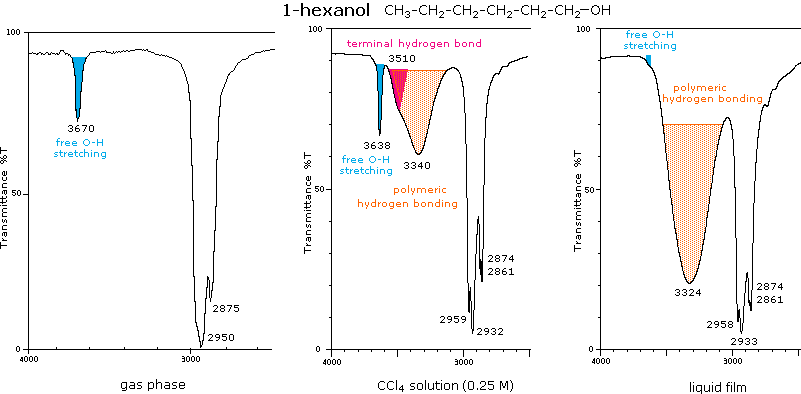
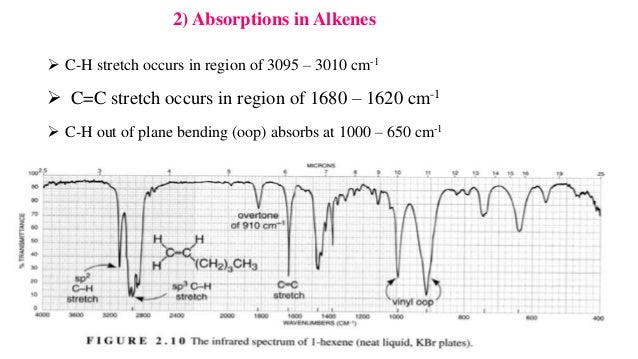
Infrared spectrometers similar in principle to the uv visible spectrometer described elsewhere permit chemists to obtain absorption spectra of compounds that are a unique reflection of their molecular structure.
Ir spectroscopy ch vinylic.
847 853 look over pages 853 866 after viewing this presentation for additional examples of various functional groups.
Vinyl alcohol ir spectra.
Nitrogen is connected to 1 carbon produces two signals one at 3350 cm 1 and the other 3450 cm 1.
Steps to analyzing an ir spectrum.
Infrared spectroscopy and mass spectrometry review of concepts fill in the blanks below.
Emphasis is on data interpretation.
833 834 in lab textbook uses of the infrared spectrum p.
Be careful to avoid focusing on selected absorption bands and overlooking others.
It is important to remember that the absence of an absorption band can often provide more information about the structure of a compound than the presence of a band.
The vinylic c h bond expected to be 3100 cm 1 3 all other c h bonds.
Each of the sentences below appears verbatim in the section.
To verify that your answers are correct look in your textbook at the end of chapter 15.
The ir spectrum table is a chart for use during infrared spectroscopy the table lists ir spectroscopy frequency ranges appearance of the vibration and absorptions for functional groups.
An example of such a spectrum is that of the flavoring agent vanillin shown below.
Look for double bonds 1600 1850cm 1 2.
Infrared spectroscopy ir theory and interpretation of ir spectra assigned readings introduction to technique 25 p.
Acyl all ir values are approximate and have a range of possibilities depending on the molecular environment in which the functional group resides.
For one thing all of these functional groups appear to the right of the c h absorptions which always occur between 2 800 cm 1 to 3 000 cm 1 in the ir spectrum and to the left of.
You can locate carbonyl groups alkenes alkynes and aromatics in the ir infrared spectrum based on their shapes and relative locations.
Use the examples linked to the table to see the profile and intensity of bands.
The method or technique of infrared spectroscopy is conducted with an instrument called an infrared.
Inductive pull of cl increases the electron density between c and o.
There are two tables grouped by frequency range and compound class.